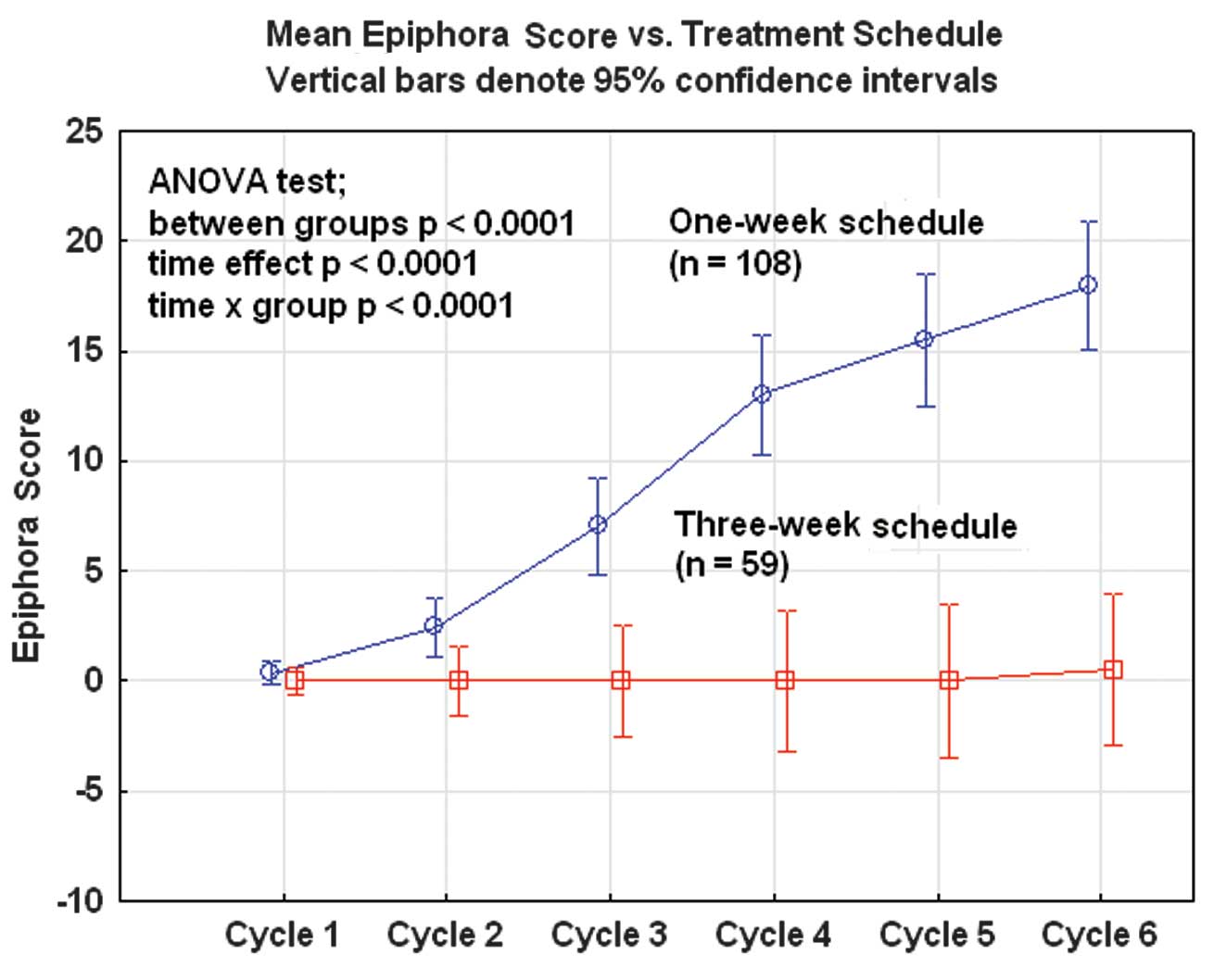
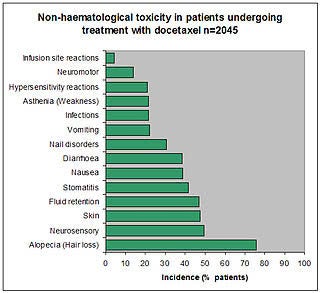
A study of docetaxel weekly or every three weeks in combination with carboplatin as first line chemotherapy in epithelial ovarian cancer: Hematological and non-hematological toxicity profiles
Administering Docetaxel for Metastatic Hormone-Sensitive Prostate Cancer 1–6 Days Compared to More Than 14 Days after the Star
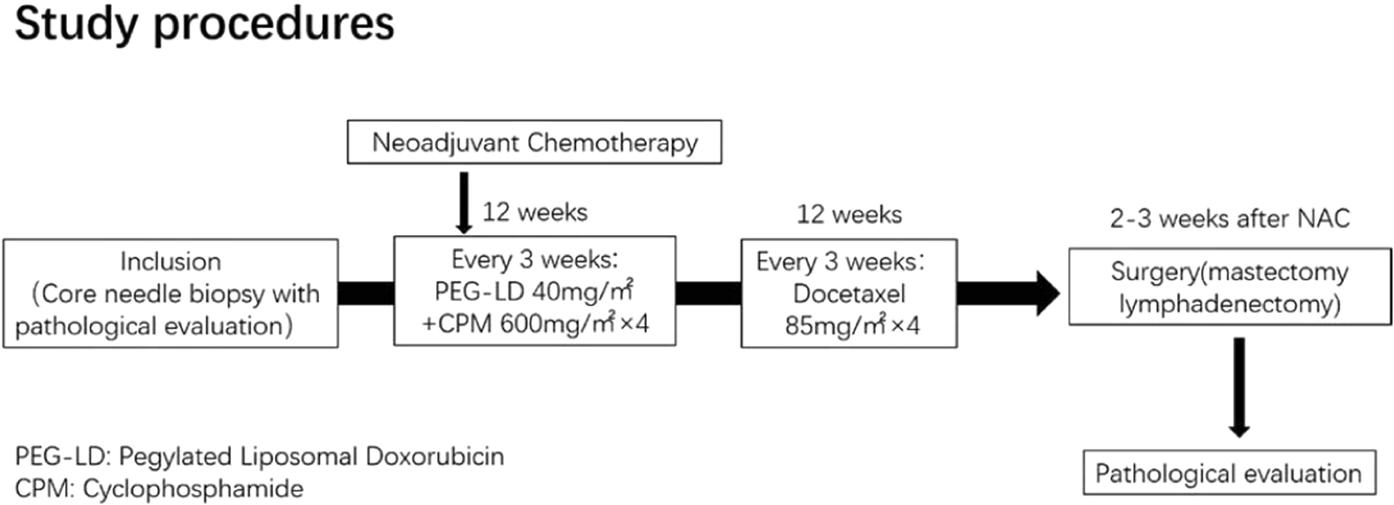
Pegylated liposomal doxorubicin plus cyclophosphamide followed by docetaxel as neoadjuvant chemotherapy in locally advanced breast cancer (registration number: ChiCTR1900023052) | Scientific Reports

Antitumoral effect and reduced systemic toxicity in mice after intra-tumoral injection of an in vivo solidifying calcium sulfate formulation with docetaxel - ScienceDirect

Taxane‐induced neurotoxicity: Pathophysiology and therapeutic perspectives - Costa - 2020 - British Journal of Pharmacology - Wiley Online Library

Management and challenges of corticosteroid therapy in men with metastatic castrate-resistant prostate cancer - Annals of Oncology

Docetaxel: An update on its molecular mechanisms, therapeutic trajectory and nanotechnology in the treatment of breast, lung and prostate cancer - ScienceDirect
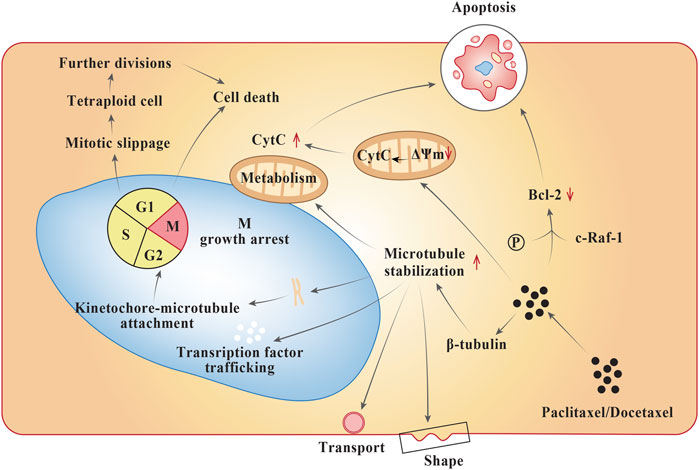
Frontiers | Platinum and Taxane Based Adjuvant and Neoadjuvant Chemotherapy in Early Triple-Negative Breast Cancer: A Narrative Review
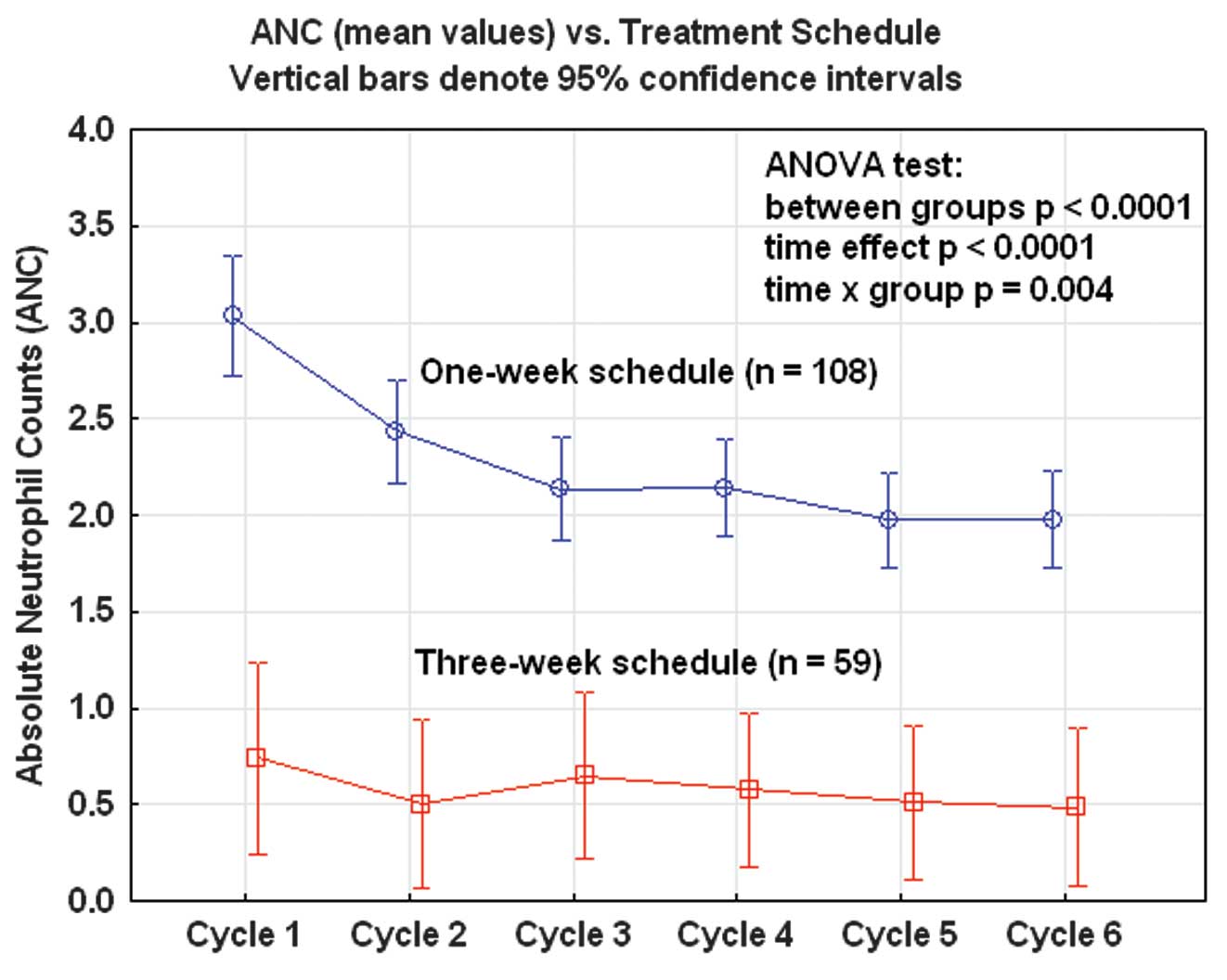
A study of docetaxel weekly or every three weeks in combination with carboplatin as first line chemotherapy in epithelial ovarian cancer: Hematological and non-hematological toxicity profiles
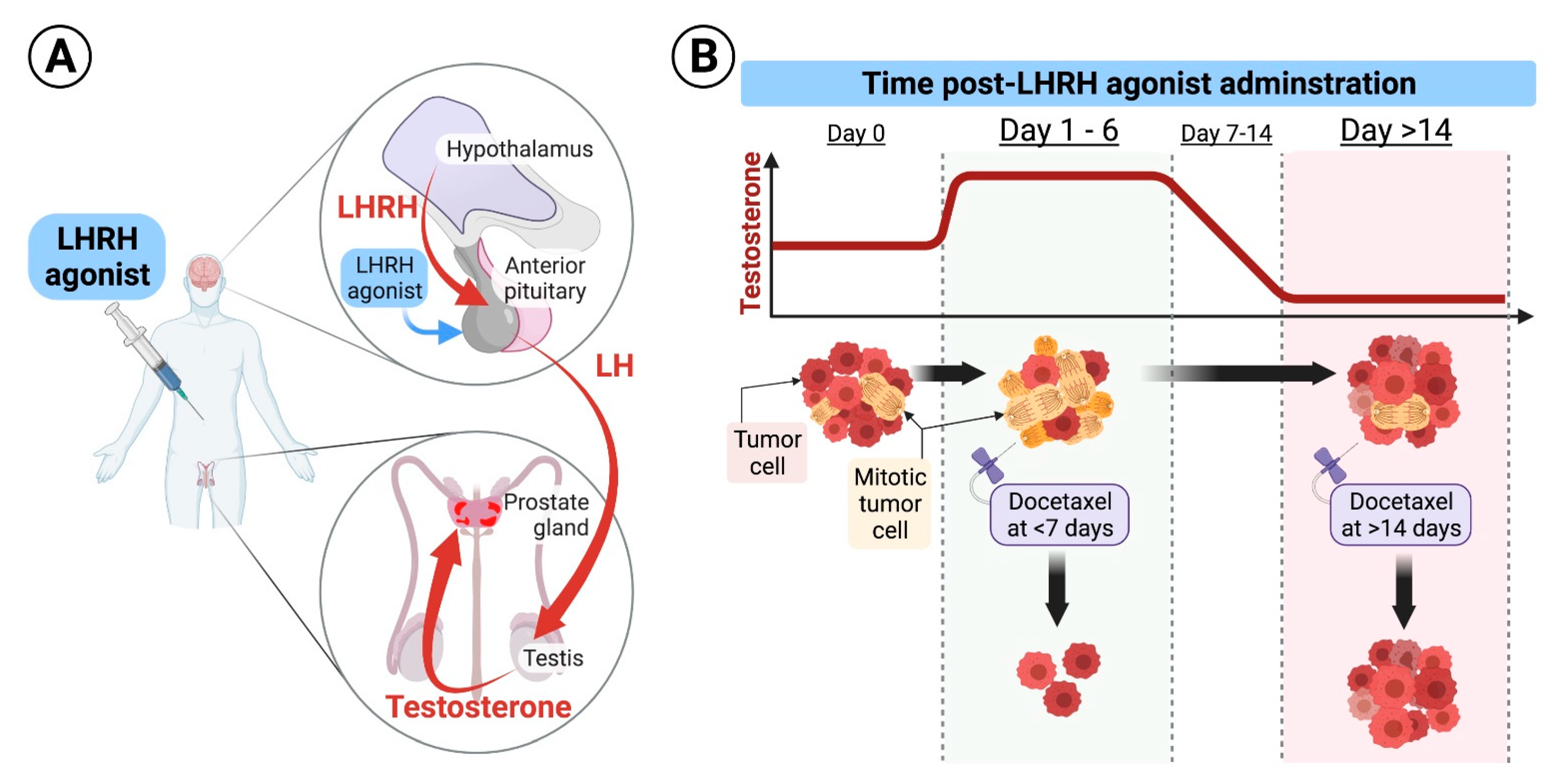
Cancers | Free Full-Text | Administering Docetaxel for Metastatic Hormone-Sensitive Prostate Cancer 1–6 Days Compared to More Than 14 Days after the Start of LHRH Agonist Is Associated with Better Clinical Outcomes









/iStock_000042502802_Large-56a5c5a03df78cf77289d952.jpg)